The distinctions between GMP plasmid DNA and research-grade plasmid DNA are critical for businesses operating in gene therapy and vaccine development. As the demand for effective therapies continues to rise, understanding these differences can significantly impact project outcomes. Companies must ensure that their plasmid DNA manufacturing process aligns with regulatory standards while meeting specific therapeutic needs.
Understanding GMP and Research-Grade Plasmid DNA
GMP (Good Manufacturing Practice) plasmid DNA is produced under strict regulatory guidelines to ensure safety, quality, and consistency. This grade is essential for clinical applications where the end product will be administered to humans. The manufacturing processes involve rigorous testing, documentation, and quality control measures, ensuring that every batch meets defined specifications.
On the other hand, research-grade plasmid DNA is typically used for laboratory experiments, preclinical studies, or initial developmental phases. These plasmids do not need to meet the stringent requirements of GMP standards, which allows for more flexibility in the production process. However, the trade-off comes in the form of variability in quality and potential impurities that could affect experimental outcomes.
Key Differences That Matter
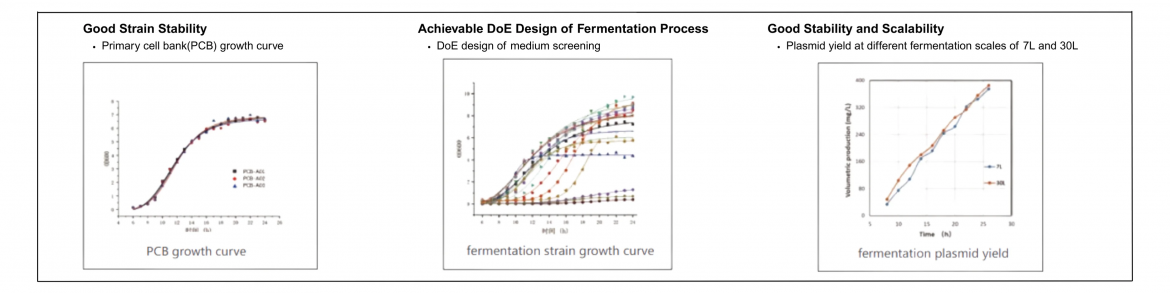
The primary distinction between GMP plasmid DNA and research-grade plasmid DNA lies in the production environment and quality assurance practices. GMP plasmid DNA manufacturing is backed by a validated quality system that encompasses everything from strain selection to fermentation processes. For instance, companies like Yaohai Bio-Pharma utilize high-quality strains with clear traceability and stable expression efficiencies, ensuring dependable results across various applications.
Research-grade products may lack this level of oversight, leading to possible inconsistencies that might compromise subsequent research efforts. With a robust plasmid manufacturing platform capable of handling fermentation scales from 7L to 2000L, GMP plasmid DNA providers can meet diverse production needs, resulting in a reliable supply chain for innovative therapies.
Moreover, GMP plasmid DNA often undergoes Quality by Design (QbD) development processes, emphasizing critical quality attributes (CQAs) and critical process parameters (CPPs). This meticulous approach enables manufacturers to ensure consistent quality across different batches. In contrast, research-grade plasmid manufacturing may bypass some of these aerated designs, limiting assurances about product performance during crucial experimental stages.
Why Choosing the Right Grade Matters
Opting for the appropriate plasmid DNA manufacturing grade can be pivotal for businesses aiming to push the boundaries of gene therapy and vaccine innovation. While research-grade plasmids might suffice for preliminary experiments, the transition to GMP plasmid DNA is crucial for advanced clinical applications. With extensive experience in CMC and clinical GMP submissions, Yaohai Bio-Pharma is well-equipped to support your journey from preclinical trials through to commercial production.
At Yaohai Bio-Pharma, they embrace a commitment to provide high-quality GMP plasmid DNA that accelerates innovation and enhances clinical trial success rates. Their antibiotic-free, animal-origin-free fermentation processes and proven track record ensure that you receive the best support for your plasmid manufacturing needs. Let them partner with you on your path toward groundbreaking therapies, ensuring you have the reliable resources necessary to drive your projects forward.